The latest 6th CAPSTONE Network Summit fulfilled the promise of an unforgettable assembly between ESRs, PIs and related partners. This time, the captivating city of Lille hosted all the provided scientific trainings and workshops, which were marvelously organized by the University of Lille, our consortium’s coordinating institution, and the local team of Pr. Rebecca Deprez-Poulain (ULILLE, U1177, Lille, FR). The scientific agenda covered a range of thematic sessions dedicated to patients and clinicians through a stimulating “round table” discussion, in-depth breakdown of clinical trials with recent examples of clinical candidates and personalized medicine, as well as enlighting lectures around the importance of ADME/PK profiling during drug discovery accompanied by an interactive hands-on workshop in experimental high-throughput methodology. This enriching experience took place at Institut Pasteur de Lille from October 18th to 20th.
Programme
|
Programme Day 1 – 18th October 2023 |
||
|
Time |
Transferable skills: Raising awareness of patient needs, clinician expectations |
Presenter |
|
9am-10am |
TABLE RONDE |
Moderators: L. Naessens-Jakovljevic (BAYER) and P. DaoPhan (BAYER) Participants : A. Folkard (Birdshot Uveitis Society), A. Cardone (Cancer Patients Europe ), D. Launay (ULILLE & Lille University Hospital) |
|
Scientific workshop:Clinical Studies |
Presenter |
|
|
10:30am-12:00pm |
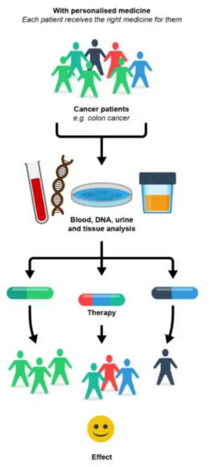
Precision medicine in oncology
|
V. Sterin (BAYER) |
|
Example of clinical development in cardiovascular disease
|
J. Mussot (BAYER) |
|
| 12:00pm-02:30pm |
ESR Poster Session 1 (Odd numbers)
|
|
| Scientific workshop:Clinical Studies | Presenter | |
| 02:30pm-03:30pm | Orphan drugs | B. Deprez (ULILLE) |
| Scientific workshop: Experimental High-Throughput methodology for screening and ADME | Presenter | |
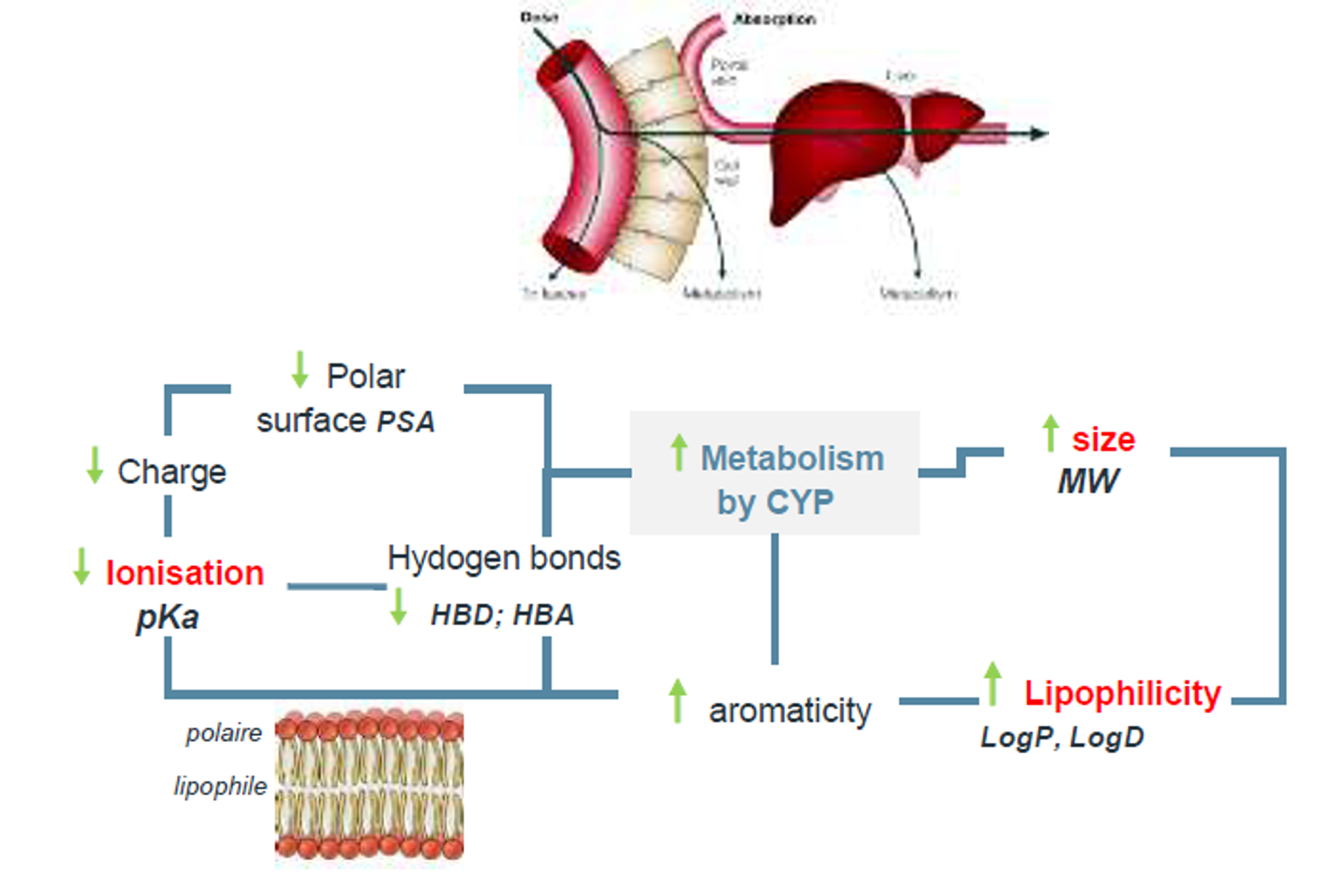
| 04:00pm-05:00pm | Principles of ADME/PK | R. Deprez-Poulain (ULILLE) |
|
Programme Day 2 – 19th October 2023 |
||
|
Time |
Scientific workshop: Clinical Studies |
Presenter |
|
9:30am-12:00pm |
Positioning and Target Product Profile |
P. Dao Phan (BAYER) |
|
Decentralized Clinical Trials |
A. Dekeyser (BAYER) |
|
|
Data Privacy |
E. Lefebvre (BAYER) | |
| 12:00pm-02:30pm | ESR Poster Session 2 (Even numbers) | |
|
Scientific workshop: Experimental HT methodology for screening and ADME |
Presenter |
|
|
2:30pm-4:00pm |
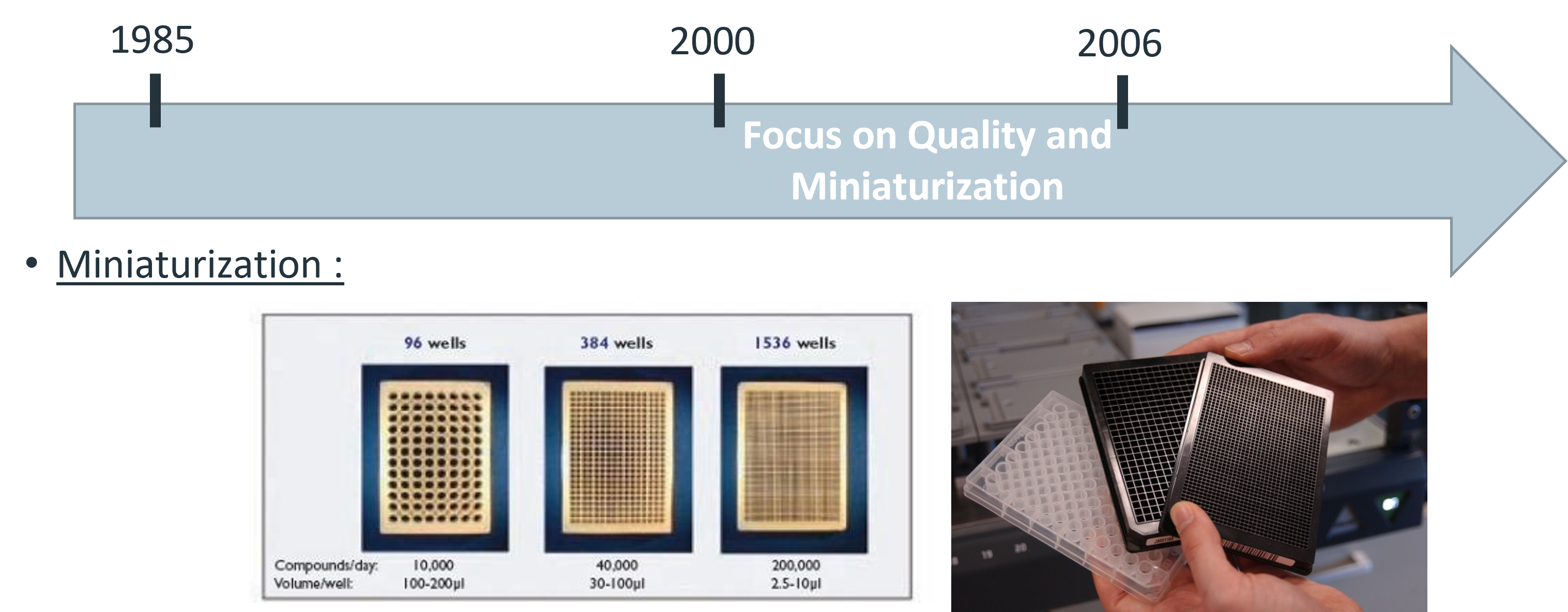
Generalities about HTS/HCS |
L. Lesire (ULILLE) |
|
Programme Day 3 – 20th October 2023 |
||
|
Time |
Scientific workshop: Experimental HT methodology for screening and ADME |
Presenter |
| 9am-10:30am | HTS/HCS in practice - Experiment on ARIADNE screening platform |
L. Lesire (ULILLE) |
| 11am-12:30pm | Pasteur Museum Visit |
R. Deprez-Poulain (ULILLE) |
| Scientific workshop: Experimental HT methodology for screening and ADME | Presenter | |
| 01:30pm-4pm | Image analysis on Columbus software |
L. Lesire (ULILLE) |
| 4pm | Poster Prizes | R. Deprez-Poulain (ULILLE) |
Details on presentations
|
 |
 |
|
|
 |
 |
|
|
 |
|
 |
 |
|
|
ESR presentations
 |
 |
During the first two days of the 6th CAPSTONE Network Summit, ESRs had the opportunity to communicate their research in a poster presentation format. ESRs presented the progress of their projects and received questions from other ESRs, PIs, and related partners. At the end of the meeting, ESRs and PIs voted for the best posters and the winners Adele Ponzoni (ESR13, Aliri, FR), Sandra Lliamas-Rizo (ESR11, NTUA, GR), Evgenia-Galateia Georgaki (ESR3, NCSRD, GR) and Alice Senni (ESR7, UPARIS, FR) received a poster prize along with a gift to remind them of the city of Lille.